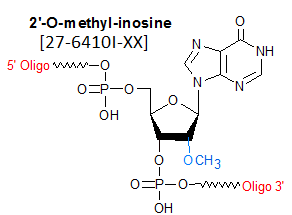
Modification : 2'-O methyl Inosine
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
| Catalog No | Scale | Price |
| 27-6410I-05 | 50 nmol | $211.00 |
| 27-6410I-02 | 200 nmol | $211.00 |
| 27-6410I-01 | 1 umol | $274.00 |
| 27-6410I-03 | 2 umol | $411.00 |
| 27-6410I-06 | 5 umol | $795.00 |
| 27-6410I-10 | 10 umol | $1,162.00 |
| 27-6410I-15 | 15 umol | $1,236.00 |
| Discounts are available for 2'-O methyl Inosine! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
Antisense Oligos (ODN) & siRNA Oligo Modifications
Click here for more information on antisense modifications, design & applications.
2'-O-Methyl inosine is classified as a 2'-O-Methyl RNA monomer. 2'-O-Methyl nucleotides are most commonly used to confer nuclease resistance to an oligo designed for anti-sense, siRNA or aptamer-based research, diagnostic or therapeutic purposes, when specific 2'-OH is not required. Nuclease resistance can be further enhanced by phosphorothiolation of appropriate internucleotide linkages within the oligo.
2'-O-Methyl bases are classified as a 2'-O-Methyl RNA monomer. 2'-O-Methyl nucleotides are most commonly used to confer nuclease resistance to an oligo designed for anti-sense, siRNA or aptamer-based research, diagnostic or therapeutic purposes, when specific 2'-OH is not required. Nuclease resistance can be further enhanced by phosphorothiolation of appropriate internucleotide linkages within the oligo.
The hydrogen bonding behavior of a 2'-O-Methyl RNA/RNA base pair is closer to that of an RNA/RNA base pair than a DNA/RNA base pair. Consequently, the presence of 2'-O-Methyl nucleotides improves duplex stability. Indeed, incorporation of a 2'-O-Methyl nucleotide into an anti-sense oligo (resulting in a 2'-O-Methyl RNA/DNA chimeric), lead to a increase in the Tm of its duplex with RNA, relative to that formed by an unmodified anti-sense DNA oligo, of 1.3oC per 2'-O-Methyl RNA residue added (2). Moreover, from a synthesis standpoint, the coupling efficiency of 2'-O-Methyl phosphoramidites are higher than those of RNA monomers, resulting in higher yield of full-length oligos.
Modifications Increasing Duplex Stability and Nuclease Resistance
|
Modification
|
Duplex Stability [Tm Increase]
|
Nuclease Resistance
|
| Locked Analog Bases |
Increased [2- 4C per substitution] |
Increased |
| 2-Amino-dA |
Increased [3.0C per substitution] |
Similar to DNA |
| C-5 propynyl-C |
Increased [2.8C per substitution] |
Increased |
| C-5 propynyl-U |
Increased [1.7C per substitution] |
Increased |
| 2'-Fluoro |
Increased [1.8C per substitution] |
Increased |
| 5-Methyl-dC |
Increased [1.3C per substitution] |
Similar to DNA |
| 2'-O Methyl |
Increased |
Increased |
| Phosphorothioate |
Slightly decreased |
Increased |
|
Click here for complete list of duplex stability modifications
|
ASO's and siRNA Modifications.
Click this link to view ASO's and siRNA Modifications.
ASO's and siRNA Delivery.
The development of effective delivery systems for antisense oligonucleotides is essential for their clinical therapeutic application. The most common delivery system involves a relatively hydrophobic molecule that can cross the lipid membrane. Cholesterol TEG, alpha-Tocopherol TEG ( a natural isomer of vitamin E), stearyl and GalNAc modifications have been shown to effective for delivery of ASO's and siRNA in addition to cell penetrating peptides.
Click this link to view these modifications.
References
1. Cotton, M.; Oberhauser, B.; Burnar, H.
et al. 2'O methyl and 2'O ethyl oligoribonucleotides as inhibitors of the in vitro U7 snRNP-dependent messenger-RNA processing event.
Nucleic Acids Res. (1991) ,
19:2629-2635.
2. Kawasaki, A.M.
et al., Uniformly modified 2'-deoxy-2'-fluoro phosphorothioate oligonucleotides as nuclease resistant antisense compounds with high affinity and specificity for RNA targets,
Journal of Medicinal Chemistry (1993),
36: 831-841.
- 2'-O methyl Inosine
