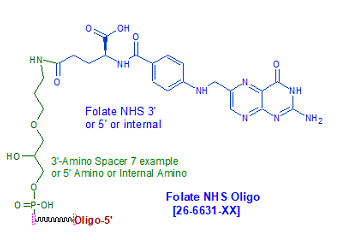
Modification : Folic Acid NHS (Vitamin B9)
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6631
Antisense & siRNA
[Folate-N]
Y
Y
Y
424.4
-
PS26-6631.pdf
-
-
-
| Catalog No | Scale | Price |
| 26-6631-05 | 50 nmol | $425.00 |
| 26-6631-02 | 200 nmol | $425.00 |
| 26-6631-01 | 1 umol | $575.00 |
| 26-6631-03 | 2 umol | $710.00 |
| 26-6631-06 | 5 umol | $935.00 |
| 26-6631-10 | 10 umol | $1,275.00 |
| 26-6631-15 | 15 umol | $1,512.00 |
Click here for a list Cellular Delivery Modifications.
Click here for more information on siRNA and antisense oligo modifications, design & applications.
Folate Oligo Modification: Vitamin B9 (Folic Acid)-Based Drug Delivery
Folic acid (FA), also known as vitamin B9, is a water-soluble vitamin involved in DNA synthesis, repair, and methylation. In recent years, FA has garnered significant attention in nanomedicine and targeted drug delivery due to its high affinity for the folate receptor (FR), which is overexpressed in various cancer cells (e.g., ovarian, breast, lung, colorectal, and kidney cancers), but minimally expressed in most normal tissues.
Mechanism of Targeting via Folate Receptor
FA binds with high affinity (Kd ≈ 10⁻⁹ M) to the folate receptor α (FR-α), a glycosylphosphatidylinositol-anchored cell surface protein. Upon binding, the FA-conjugated drug delivery system is internalized by receptor-mediated endocytosis, leading to the release of the drug inside the target cells. This mechanism enhances selectivity and reduces off-target toxicity in chemotherapy
Advantages of Folic Acid in Targeted Delivery
High receptor binding specificity
Small size and chemical stability
Non-immunogenic
Easy chemical conjugation
Effective targeting of various solid tumors
References
1. Liu Y, et al. (2007). Folate receptor-targeted delivery of siRNA using PEG-PEI. Mol Pharmaceutics, 4(5):695-705.
2. Chen Y, et al. (2010). Tumor-targeted siRNA delivery via folate-receptor mediated endocytosis. Biomaterials, 31(3):524-537.
3. Zhang Y, et al. (2012). Folic acid-chitosan nanoparticles for siRNA delivery to breast cancer. Int J Nanomedicine, 7:2227-2238.
4. Kim J, et al. (2010). Multifunctional gold nanoparticles for siRNA delivery and imaging. ACS Nano, 4(7):3689-3696.
5. Kesharwani P, et al. (2015). Dendrimer-based targeted delivery of siRNA. Drug Discov Today, 20(5):536-547.
siRNA Oligo Cellular Delivery Modifications
Vitamin B12 (Cyanocobalamin) Oligo
The vitamin B12-Cobalmine conjugated oligo may help in cellular delivery of siRNA to the brain and nervous system. Cyanocobalamin (commonly known as Vitamin B12) is a highly complex, essential vitamin, owing its name to the fact that it contains the mineral, cobalt. This vitamin is produced naturally by bacteria, and is necessary for DNA synthesis and cellular energy production. Vitamin B12 has many forms, including the cyano-, methyl-, deoxyadenosyl- and hydroxy-cobalamin forms. The cyano form, is the most widely used form in supplements and prescription drugs.
Vitamin B12-NHS Ester is a derivative of Vitamin B12 which is a water-soluble vitamin with a key role in the normal functioning of the brain and nervous system, and for the formation of blood. The NHS ester is utilized to conjugate to amino derivatized oligo.
Vitamin E. alpha-tocopherol TEG Modification
alpha-tocopherol (vitamin E) is both lipophilic and non-toxic even at high doses so would be an excellent candidate as a lipophilic carrier for oligonucleotides. Similar to cholesterol TEG, the TEG liker arm facilitates solubility issues of the oligo making it soluble in aqueous buffers.
Cholesterol TEG Modification
Cholesterol TEG Modification is a lipophilic modification aiding in cellular delivery. The TEG liker arm facilitates solubility issues of the oligo making it soluble in aqueous buffers.
Stearyl Modification
Stearyl Modification is C18 lipid, it is an economical and effective carrier molecule. We envisage that the 5'-stearyl group will become a favored lipophilic carrier for experimentation with synthetic oligonucleotides.
GalNAc Trivalent Modification
A more directed approach to the delivery of therapeutic oligonucleotides specifically to the liver has been to target the asialoglycoprotein receptor (ASGPR) using a suitable glycoconjugate. Indeed, ASGPR is the ideal target for delivery of therapeutic oligonucleotides to the liver since it combines tissue specificity, high expression levels and rapid internalization and turnover. The use of oligonucleotide glycoconjugates has led to significant advances in therapeutic delivery as evidenced by the work of Alnylam Pharmaceuticals which has developed multivalent N-acetylgalactosamine (GalNAc) conjugated siRNAs that bind at nanomolar levels to ASGPR (1). A similar strategy has been applied at Ionis Pharmaceuticals directed at the development of antisense oligonucleotide therapeutics (2).
The GalNAc ligand originally used by Alnylam is the triantennary ligand would seem to lend itself to formation by post synthesis conjugation to the 3' terminus but a complex trivalent GalNAc support would also be perfectly applicable, if challenging to produce. However, an alternative approach using a monovalent GalNAc support with two additions of a monovalent GalNAc phosphoramidite was also described and yielded a trivalent GalNAc structure. This (1+1+1) trivalent GalNAc structure led to GalNAc modified siRNA oligos with potency equal to the equivalent siRNA with the triantennary GalNAc ligand both in vitro and in vivo.
Researchers at Ionis have developed antisense oligonucleotides containing the GalNAc cluster. In their case, they were able to show2 that moving the triantennary GalNAc ligand to the 5' terminus led to improved potency in vitro and in vivo. As may be expected, such a large complex ligand lends itself to solution phase chemistry to produce GalNAc modified antisense oligos. However, a solid phase synthetic approach was also described, and compared to the solution phase approach structure of the 5'-GalNAc triantennary ligand (4).
A further report on antisense oligonucleotides demonstrated (5) the effectiveness of modifying at the 5' terminus using monovalent GalNAc ligands. Up to five GalNAc monomers were added in a serial manner (Figure 3) and it was shown that activity of the antisense oligonucleotides improved as the number of GalNAc units increased. The authors also showed that phosphodiester linkages between the GalNAc units were preferable to phosphorothioate linkages in their testing (5).
References. Adapted from Glen Research Reports. http://www.glenresearch.com/GlenReports/GR29-14.html
1. J.K. Nair, et al., J Am Chem Soc, 2014, 136, 16958-61.
2. T.P. Prakash, et al., Bioorganic & Medicinal Chemistry Letters, 2015, 25, 4127-4130.
3. K.G. Rajeev, et al., Chembiochem, 2015, 16, 903-8.
4. I. Cedillo, et al., Molecules, 2017, 22.
5. T. Yamamoto, M. Sawamura, F. Wada, M. Harada-Shiba, and S. Obika, Bioorganic & Medicinal Chemistry, 2016, 24, 26-32.
- Folic Acid NHS (Vitamin B9)
