In this section: Introduction | Quality Control | Purification | Modifications | Long Oligos | Price List
In this section: Introduction | Molecular Beacon FAQ's | Fluorescent Probes Price List | Other Fluorescent Molecular Probes
In this section: SPCT | DME SPCT Intro | Order DME TaqMan® Assays SPCT | SNP PCT Search | Gene Expression Assays | SPCT Design Center | GeneAssays
In this section: RNA Oligonucleotides | Quality Control | Purification | Modifications | RNAi Explorer™ Products and Prices | Custom RNAi | RNAi Design Guidelines | SmartBase™ siRNA Modifications | shRNA Explorer™
In this section: PCR Amplification & Analysis
In this section: Introduction | Genemer™ | GeneProber™ | Prober™ Gene Detection Kits | GScan™ Gene Detection Kits | Genemer™ Control DNA | Infectious Diseases
In this section: Gene Construction
In this section: Introduction | The Omni-Clean™ System | The Omni-Pure™ Plasmid Purification System | The Omni-Pure™ Genomic DNA Purification System | Viral DNA & RNA Purification | Microbial DNA Purification | Plant DNA Purification
In this section: Introduction | Quality Control | Purification | Modifications | Long Oligos | Price List
In this section: Introduction | Molecular Beacon FAQ's | Fluorescent Probes Price List | Other Fluorescent Molecular Probes
In this section: SPCT | DME SPCT Intro | Order DME TaqMan® Assays SPCT | SNP PCT Search | Gene Expression Assays | SPCT Design Center | GeneAssays
In this section: RNA Oligonucleotides | Quality Control | Purification | Modifications | RNAi Explorer™ Products and Prices | Custom RNAi | RNAi Design Guidelines | SmartBase™ siRNA Modifications | shRNA Explorer™
In this section: PCR Amplification & Analysis
In this section: Introduction | Genemer™ | GeneProber™ | Prober™ Gene Detection Kits | GScan™ Gene Detection Kits | Genemer™ Control DNA | Infectious Diseases
In this section: Gene Construction
In this section: Introduction | The Omni-Clean™ System | The Omni-Pure™ Plasmid Purification System | The Omni-Pure™ Genomic DNA Purification System | Viral DNA & RNA Purification | Microbial DNA Purification | Plant DNA Purification
DBCO-Maleimide
DBCO-Maleimide
Code : [DBCO-Mal]
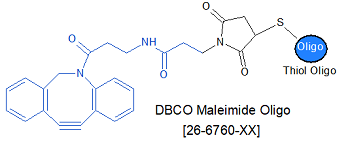
Modification : DBCO-Maleimide
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6760
Click Chemistry
[DBCO-Mal]
Y
Y
Y
427.4
-
PS26-6760.pdf
-
-
-
| Catalog No | Scale | Price | | 26-6760-05 | 50 nmol | $308.00 | | 26-6760-02 | 200 nmol | $308.00 | | 26-6760-01 | 1 umol | $394.00 | | 26-6760-03 | 2 umol | $513.00 | | 26-6760-06 | 5 umol | $785.00 | | 26-6760-10 | 10 umol | $1,348.00 | | 26-6760-15 | 15 umol | $1,685.00 |
Click here for a complete list of Click Chemistry Oligo Modifications
This modification is a post synthesis maleimide conjugation to a reduced thiol amino group thus an additional modification with thiol group is required. A C3 or C6 thiol group can be placed at the 5' or for internal positions Thiol C6 dT modified base is used.
Cyclooctyne-based (dibenzocyclooctynes, DBCO) modifications offers the ease of copper-free click reagents. These are simple to use and has excellenet click performance in 17 hours or less at room temperature. Gene Link offers 5'-DBCO-TEG for preparing oligos with 5'-DBCO and a 15 tom triethylene glycol spacer arm, DBCO-dT for inserting a DBCO group at any position within the oligonucleotide and DBCO-sulfo-NHS Ester is also offered for post-synthesis conjugation reactions. DBCO-modified oligos may be conjugated with azides in organic solvents, such as DMSO, or aqeous buffers. Depending on the azide used, the reaction will go to completion in 4-17 hours at room temperature.
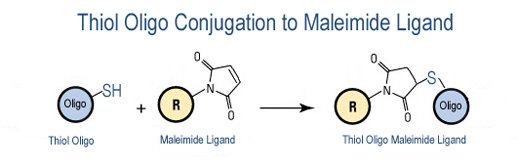
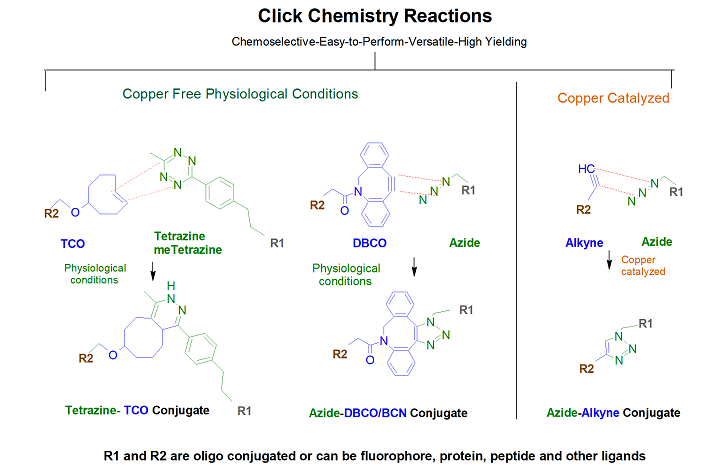
- DBCO-Maleimide
|
|
