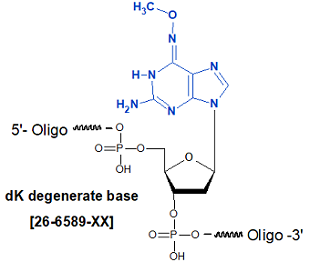
Modification : dK degenerate base
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6589
Duplex Stability
[dK]
Y
Y
Y
358.25
7.7
PS26-6589.pdf
-
-
-
| Catalog No | Scale | Price |
| 26-6589-05 | 50 nmol | $254.00 |
| 26-6589-02 | 200 nmol | $254.00 |
| 26-6589-01 | 1 umol | $288.00 |
| 26-6589-03 | 2 umol | $335.00 |
| 26-6589-06 | 5 umol | $1,296.00 |
| 26-6589-10 | 10 umol | $728.00 |
| 26-6589-15 | 15 umol | $899.00 |
| Discounts are available for dK degenerate base! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
Click here for a complete list of Degenerate Bases & Spiking Oligo Modifications
The degeneracy of the genetic code complicates the design of primers in the absence of unique sequence information. Classically inosine has been used at degenerate position as it is able to base pair with all four bases (1) and function as a universal base. Similarly 5-nitroindole was determined to be an effective universal base (2). The modified bases designated P and K show considerable promise as degenerate bases. The pyrimidine derivative P, when introduced into oligonucleotides, base pairs with either A or G, while the purine derivative K base pairs with either C or T. A dP+dK mix also can serve as a mixed base with much less degeneracy than dA+dC+dG+dT (N) (3, 4).
The degenerate bases P and K are copied as either pyrimidine (C or T) or either purine (A or G), respectively. They can be used together in oligomers to prime DNA synthesis in PCR and in sequencing reactions. In a direct comparison, they were more effective in primers than dI.
Fully degenerate sites may be formed in an oligonucleotide using an A/C/G/T mix but the complexity of the mixture of oligonucleotides formed in this way obviously rises with each insertion and limits the usefulness of this technique. The pyrimidine derivative P, when introduced into oligonucleotides, base pairs with either A or G , while the purine derivative K base pairs with either C or T. Oligonucleotides containing one or more P substitutions were found to form duplexes of stability equivalent to the parent sequence and exhibited sharp transitions on melting. Substitution with one or more K residues led to duplexes of reduced but still effective stability (3, 4).
Recommended Further Reading
Glen Report 8.1: New Universal and Degenerate Bases.
Glen Report 9.1: Properties of Oligonucleotides Containing the Bases P and K.
References:
1. Liu, H. and Nichols, R. PCR amplification using deoxyinosine to replace entire codon and at ambiguous positions.
Biotechniques. (1994),
16: 24-26.
2. Loakes, D. and Brown, D.M. 5-Nitroindole as a universal base analogue. Nucleic Acids Res. (1994), 22: 4039-4043.
3. Lin, P.K. and Brown, D.M. Synthesis and duplex stability of oligonucleotides containing cytosine-thymine analogues. Nucleic Acids Res. 1989 Dec 25;17(24):10373–10383
4. Lin, P.K. and Brown, D.M. Synthesis of oligodeoxyribonucleotides containing degenerate bases and their use as primers in the polymerase chain reaction. Nucleic Acids Res., (1992) 20: 5149-5152.
- dK degenerate base
