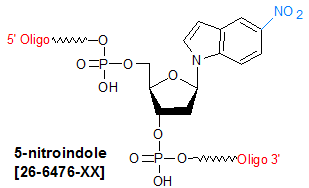
Modification : 5-nitroindole
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6476
Duplex Stability
[5NitInd]
Y
Y
Y
340.23
16
PS26-6476.pdf
-
-
-
| Catalog No | Scale | Price |
| 26-6476-05 | 50 nmol | $162.00 |
| 26-6476-02 | 200 nmol | $162.00 |
| 26-6476-01 | 1 umol | $211.00 |
| 26-6476-03 | 2 umol | $316.00 |
| 26-6476-06 | 5 umol | $949.50 |
| 26-6476-10 | 10 umol | $1,685.00 |
| 26-6476-15 | 15 umol | $2,106.00 |
| Discounts are available for 5-nitroindole! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
Click here for a complete list of Degenerate Bases &
Spiking Oligo Modifications
5-Nitroindole is a hydrophobic aromatic compound, and can be used as a universal base analog in oligonucleotides. The term "universal base" refers to a base with the ability to replace any of the four natural bases without significantly destabilizing either neighboring base-pair interactions or disrupting the expected functional capability of the resulting modified oligonucleotide. Incorporation of universal bases into oligos is desirable in cases when either imprecise or random base-pairing is required, and the resulting "mis-matched" complements need to be stable. Examples of such situations include reverse-translation of known protein sequence for oligo design (oligos to be used as primers or probes), development of an in vitro or in vivo oligo probe able to hybridize to related but distinct genes (for example, viral sub-strains or allelic variants--SNPs, indels, etc.), in vitro mutagenesis and motif cloning.
5-Nitroindole does not discriminate between the four natural nucleotide bases in duplex formation. This is in contrast to deoxyinosine (dI), which, although often used as a "universal" base, actually shows fairly strong base-pair bias. 5-Nitroindole's lack of discrimination ("universality") is a consequence of the fact that it does not form hydrogen bonds with natural bases. Instead, 5-nitroindole stabilizes the duplex via base-stacking interactions, and is superior in this regard than 3-nitropyrrole, which also has been used as a universal base (1,2). Much research work on 5-nitroindole has focused on its potential as an alternative to the used of mixed bases in PCR or Sanger sequencing primers. The ability of 5-nitroindole to function in this way, however, is strongly dependent on where it is located in the primer. Based on the results of several research studies (reviewed in 3), the following guidelines may be useful in optimizing the utility of this base analog for these applications:
1. Substitution of 5-nitroindole as a universal base is less destabilizing towards the ends of oligos than towards the center.
2. Grouped substitutions are more easily tolerated than spaced ones, that is, contiguous rather than codon third substitutions.
3. If
more than two codon third substitutions are incorporated into a PCR/sequencing primer, priming generally is poor.
4. Up to four contiguous substitutions in the middle or 5'-end of a PCR/sequencing primer generally yield acceptable amounts of PCR product or sequencing ladder. Above that priming is poor, due to the run of 5-nitroindoles forming undesirable secondary structures in the primer.
5. Substitutions at, or within eight bases of, the 3'-end produce primers that generally are ineffective at priming.
Beyond primers, nested sets of universal oligonucleotide probes containing 5-nitroindole have been successfully used to target regions of rRNA in a variety of microorganism species, in order to ensure equal probe specificity in all target organisms (4). 5-nitroindole has also been used to examine protein-DNA interactions relevant to nucleotide excision repair (NER) (5) and RNA polymerase binding (6). In addition, the potential of 5-nitroindole to enhance the stability of duplexes formed between a target DNA and octa-/decanucleotide probes immobilized on DNA microarrays has been investigated (7). Finally, 5-nitroindole often has been used to stabilize DNA structures of different types (3).
References
1. Loakes, D.; Brown, D.M. 5-Nitroindole as a universal base analogue.
Nucleic Acids Res. (1994),
22: 4039-4043.
2. Loakes, D.; Hill, F.; Brown, D.M.; Salisbury, S.A. Stability and structure of DNA oligonucleotides containing non-specific base analogues.
J. Mol. Biol. (1997),
270: 426-435.
3. Loakes, D. The applications of universal DNA base analogues.
Nucleic Acids Res. (2001),
29: 2437-2447.
4. Zheng, D.; Raskin, L. Quantification of
Methanosaeta species in anaerobic bioreactors using genus- and species-specific hybridization probes.
Microb. Ecol. (2000),
39: 246-262.
5. Buschta-Hedayat, N.; Buterin, T.; Hess, M.T.; Missura, M.; Naegeli, H.Recognition of non-hybridizing base pairs during nucleotide excision repair of DNA.
Proc. Natl Acad. Sci. USA (1999),
96: 6090-6095.
6. Matlock, D.L.; Heyduk, T. Sequence determinants for the recognition of the fork junction DNA containing the -10 region of promoter DNA by E. coli
RNA polymerase
Biochemistry (2000),
39: 12274-12283.
7. Parinov, S.; Barsky, V.; Yershov, G.; Kirillov, E.; Timofeev, E.; Belgovskiy, A.; Mirzabekov, A. DNA sequencing by hybridization to microchip octa- and decanucleotides extended by stacked pentanucleotides.
Nucleic Acids Resl. (1996),
24: 2998-3004.
- 5-nitroindole
