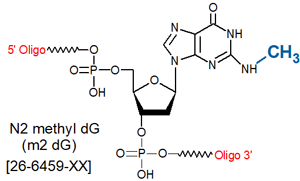
Modification : N2-Methyl dG (m2dG)
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6459
Structural Studies
[m2dG]
Y
Y
Y
343.24
11.5
PS26-6459.pdf
-
-
-
| Catalog No | Scale | Price |
| 26-6459-05 | 50 nmol | $885.00 |
| 26-6459-02 | 200 nmol | $885.00 |
| 26-6459-01 | 1 umol | $912.00 |
| 26-6459-03 | 2 umol | $1,265.00 |
| 26-6459-06 | 5 umol | $3,379.50 |
| 26-6459-10 | 10 umol | $6,588.00 |
| 26-6459-15 | 15 umol | $7,452.00 |
| Discounts are available for N2-Methyl dG (m2dG)! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
Guanine bases in DNA are susceptible to N-alkylation by various carcinogens, leading to miscoding and mutagenicity. Choi and Guengerich (1) have prepared a series of N2-alkyl-2’- deoxyguanosine phosphoramidites where the alkyl group ranges in size from methyl to anthracenylmethyl for studies on the effect of the size of these groups on the catalytic efficiency and fidelity of various DNA polymerases (1). Gene Link offers N2-methyl-dG [26-6459], N2-ethyl-dG [26-6585] N2-benzyl dG [26-6586] and N2-isobutyl-dG [26-6587] modifications for probing the steric requirements at N2 of dG in various applications.
N2-Methyl-deoxyguanosine (N2-Me-dG, m2 dG) is a methylated nucleoside base, and is primarily used in the study of DNA damage and repair mechanisms related to alkylation damage. The N1-Me-dA lesion is primarily generated by SN2 alkylating reagents such as methyl methanesulfonate and dimethylsulfate, which react with the N1 position of adenine (2). In cells, N1-methyl-dA acts as a lethal DNA replication block, but is not very mutagenic (1% A to T transversion in E. coli), and is repaired by the enzyme AlkB by direct reversal (3,4). Because the N1 position of adenine is involved in hydrogen bonding of A : T Watson-Crick base pairing, methylation of this site was expected to disrupt hydrogen bonding. However, NMR analysis revealed that N1-methylation actually alters the A:T base-pairing interactions from Watson-Crick to (syn)N1-methyl-A : (anti)T Hoogsteen, thus providing insight into why AlkB repair of N1-Methyl-dA lesions is 10X more efficient on ssDNA over dsDNA (5).
References
(1) Choi, J.-Y.; Guengerich, F. P.
J. Biol. Chem. 2004, 279 , 19217-19229.
(1) Sedgwick, B., Lindahl, T. Recent progress on the Ada response for inducible repair of DNA alkylation damage.
Oncogene (2002),
21: 8886-8894.
(2) Chen, B.J., Carroll, P., Samson, I. The Eschericia coli alkB protein protects human cells against alkylation-induced toxicity.
J. Bacteriol. (1994),
176: 6255-6261.
(3) Delaney, J.C., Essigman, J.M. Mutagenesis, genotoxicity and repair of 1-methyladenine, 3-alkylcytosines, 1-methylguanine, and 3-methylthymine in alkB Escherichia coli.
Proc. Natl. Acad. Sci. (USA) (2004),
101: 14051-14056.
(4) Yang, H., Zhan, Y., Fenn, D., Chi, L.M., Lam, S.K. Effect of 1-methyladenine on double-helical DNA structures.
FEBS Letters (2008),
582: 1629-1633.
- N2-Methyl dG (m2dG)
