Modification : Spacer 9
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
| Catalog No | Scale | Price |
| 26-6440-05 | 50 nmol | $138.00 |
| 26-6440-02 | 200 nmol | $138.00 |
| 26-6440-01 | 1 umol | $180.00 |
| 26-6440-03 | 2 umol | $269.00 |
| 26-6440-06 | 5 umol | $810.00 |
| 26-6440-10 | 10 umol | $1,438.00 |
| 26-6440-15 | 15 umol | $1,797.00 |
| Discounts are available for Spacer 9! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
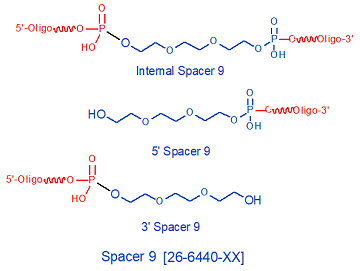
Spacer 9 is a triethylene glycol (PEG30 chain that is 9 atoms long (6 carbons + 3 oxygens), and is used to incorporate a spacer arm into an oligonucleotide. Spacer 9 can be incorporated in consecutive additions whenever a longer spacer is required. Spacer 9 has been used to form non-nucleotide bridges in hairpin loops in oligonucleotides (1), for linking oligonucleotides to epitopes for drug development (2), and for solid-phase immobilization of hybridization probes (3). Multiple incorporation of Spacer 9 has been used to form long, flexible linker arms between the two domains (double-helix forming and triple-helix forming, respectively) of a bifunctional DNA oligonucleotide, in order to maximize the binding flexibility of the two domains for their respective targets (4). This oligo was used to form a peptide nucleic acid (PNA)-DNA conjugate for use in site-directed recombination applications.
Gene Link offers spacers of various length, examples C2, C3, C6, C12 and Spacer 9 and 18. These can be inserted multiple times to yield a total much longer spacer length.
References
1. Nelson, J.S., Giver, L., Ellington, A.D., Letsinger, R.L. Incorporation of Non-Nucleotide Bridge into Hairpin Oligonucleotides Capable of High-Affinity Binding to the Rev Protein of HIV-1.
Biochemistry. (1996),
35: 5339-5344.
2. Palma, E., Klapper, D.G., Cho, M.J. Antibodies as Drug Carriers III: Design of Oligonucleotides with Enhanced Binding Affinity for Immunoglobulin G.
Pharm. Res. (2005),
22: 122-127.
3. Beattie, W.G., Meng, L., Turner, S.L., Varma, R.S., Dao, D.D., Beattie, K.L. Hybridization of DNA targets to glass-tethered oligonucleotide probes.
Mol. Biotechnol. (1995),
4: 213-225.
4. Rogers, F.A., Vasquez, K.M., Egholm, M., Glazer, P.M. Site-directed recombination via bifunctional PNA-DNA conjugates.
Proc. Natl. Acad. Sci. USA (2002),
99: 16695-16700.
Oligonucleotide PEGylation : Spacers vs. PEGylation
Gene Link offers short PEG3 and PEG6 as direct coupling using automated chemistry. The PEG3 is termed as Spacer 9 and PEG6 as spacer 18. These are also used to introduce space between adjacent sequence and modifications. These can be inserted multiple times to increase the PEG units.
Larger 2, 5, 10 and 20 kDa PEGylation of oligonucleotides is inserted at any site of an oligonucleotide using a post synthesis amino group on the oligo with PEG-NHS.
PEGylation is the covalent attachment of polyethylene glycol (PEG) to oligonucleotides such as DNA, RNA, antisense, siRNA and aptamers. It improves pharmacokinetics, reduces renal clearance, increases nuclease stability, and decreases immunogenicity. (1) The way PEG shields its conjugated payload offers new challenges and opportunities for oligonucleotide PEGylation. Other than aptamers, the target of most oligonucleotides is a complementary sequence.
Comparison of PEGylation Size & Biological Outcome
|
PEG Size (Ethylene Glycol Units)
|
Hydrodynamic Effect
|
Typical Outcome
|
| 2 kDa (~44) |
Minimal |
Slight stability increase |
| 5 kDa (~114) |
Moderate |
Partial half-life improvement |
| 10 kDa (~227) |
Strong |
Reduced renal clearance |
| 20 kDa (~455) |
Very strong |
Long circulation |
| 40 kDa (~910) |
Extreme |
Depot-like behavior |
|
Pharmacokinetics depend on hydrodynamic diameter, not molecular weight of the oligo.
|
|
Unmodified 20-mer oligo ≈ ~7 kDa. Kidney filtration cutoff ≈ 40-60 kDa hydrodynamic equivalent
|
|
Oligo PEG size controls circulation time versus tissue penetration. Optimal design balances exposure and activity
|
Messenger RNA (mRNA) delivery strategies are required to protect biologically fragile
mRNA from ribonuclease (RNase) attacks to achieve efficient therapeutic protein expression. To tackle this issue, most mRNA delivery systems have used cationic components.
A cation-free delivery strategy by hybridization of PEGylated RNA oligonucleotides with mRNA. The PEG strands on the mRNA sterically and electrostatically shields the mRNA, improving mRNA
nuclease stability 15-fold and the PEGylated mRNA induced nearly 20-fold higher efficiency of reporter protein expression than unhybridized mRNA in cultured cells (2).
PEGylation has been used to improve the biopharmaceutical properties of protein drugs since the 1990s, and over a dozen PEGylated pharmaceuticals are currently on the market (2). PEG creates a large hydration shell, which sterically blocks other biomacromolecules from penetrating through the polymer layer and binding with the interior substrate (3, 4). Binding requires displacing the PEG by the incoming molecule, generally making such binding less thermodynamically favorable. These properties usually result in weaker interactions between the receptor and the conjugated molecule, but increased drug solubility, prolonged blood circulation, and increased drug stability often offset by the reduced binding affinity. PEGylated oligonucleotides can be an exception to this generalization, with increased binding to a complementary sequence compared to unmodified ONs. The effect is attributed to macromolecular volume exclusion (6).
PEGylation References
1. Li WJ; Zhan P; De Clercq E; Lou HX; Liu XY Current drug research on PEGylation with small molecular agents. Prog. Polym. Sci 2013, 38, 421-444.
2. Yoshinaga, N; Naito, M; Tachihara, Y; Boonstra, E; Osada, K; Cabral, H and Uchida, S. PEGylation of mRNA by Hybridization of Complementary PEG-RNA Oligonucleotides Stabilizes mRNA without Using Cationic Materials. Pharmaceutics 2021, 13, 800.
3. Harris JM; Chess RB Effect of pegylation on pharmaceuticals. Nat. Rev. Drug Discov 2003, 2, 214-221. [PubMed: 12612647]
4. Harris JM; Martin NE; Modi M Pegylation. Clin. Pharmacokinet 2001, 40, 539-551. [PubMed: 11510630]
5. Plesner B; Fee CJ; Westh P; Nielsen AD Effects of PEG size on structure, function and stability of PEGylated BSA. Eur. J. Pharm. Biopharm 2011, 79, 399-405. [PubMed: 21620970]
6. Nakano S-I; Karimata H; Ohmichi T; Kawakami J; Sugimoto N The effect of molecular crowding with nucleotide length and cosolute structure on DNA duplex stability. J. Am. Chem. Soc 2004, 126, 14330-14331. [PubMed: 15521733]
- Spacer 9
