In this section: Introduction | Quality Control | Purification | Modifications | Long Oligos | Price List
In this section: Introduction | Molecular Beacon FAQ's | Fluorescent Probes Price List | Other Fluorescent Molecular Probes
In this section: SPCT | DME SPCT Intro | Order DME TaqMan® Assays SPCT | SNP PCT Search | Gene Expression Assays | SPCT Design Center | GeneAssays
In this section: RNA Oligonucleotides | Quality Control | Purification | Modifications | RNAi Explorer™ Products and Prices | Custom RNAi | RNAi Design Guidelines | SmartBase™ siRNA Modifications | shRNA Explorer™
In this section: PCR Amplification & Analysis
In this section: Introduction | Genemer™ | GeneProber™ | Prober™ Gene Detection Kits | GScan™ Gene Detection Kits | Genemer™ Control DNA | Infectious Diseases
In this section: Gene Construction
In this section: Introduction | The Omni-Clean™ System | The Omni-Pure™ Plasmid Purification System | The Omni-Pure™ Genomic DNA Purification System | Viral DNA & RNA Purification | Microbial DNA Purification | Plant DNA Purification
In this section: Introduction | Quality Control | Purification | Modifications | Long Oligos | Price List
In this section: Introduction | Molecular Beacon FAQ's | Fluorescent Probes Price List | Other Fluorescent Molecular Probes
In this section: SPCT | DME SPCT Intro | Order DME TaqMan® Assays SPCT | SNP PCT Search | Gene Expression Assays | SPCT Design Center | GeneAssays
In this section: RNA Oligonucleotides | Quality Control | Purification | Modifications | RNAi Explorer™ Products and Prices | Custom RNAi | RNAi Design Guidelines | SmartBase™ siRNA Modifications | shRNA Explorer™
In this section: PCR Amplification & Analysis
In this section: Introduction | Genemer™ | GeneProber™ | Prober™ Gene Detection Kits | GScan™ Gene Detection Kits | Genemer™ Control DNA | Infectious Diseases
In this section: Gene Construction
In this section: Introduction | The Omni-Clean™ System | The Omni-Pure™ Plasmid Purification System | The Omni-Pure™ Genomic DNA Purification System | Viral DNA & RNA Purification | Microbial DNA Purification | Plant DNA Purification
Digoxigenin NHS
Digoxigenin NHS
Code : [Dig-N]
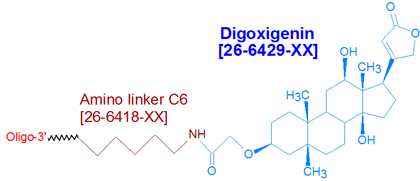
Modification : Digoxigenin NHS
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6429
Affinity Ligands
[Dig-N]
Y
Y
Y
561.3
13
PS26-6429.pdf
-
-
-
| Catalog No | Scale | Price | | 26-6429-05 | 50 nmol | $650.00 | | 26-6429-02 | 200 nmol | $650.00 | | 26-6429-01 | 1 umol | $1,250.00 | | 26-6429-03 | 2 umol | $1,875.00 | | 26-6429-06 | 5 umol | $4,797.00 | | 26-6429-10 | 10 umol | $6,951.00 | | 26-6429-15 | 15 umol | $8,864.00 |
This modification is a post synthesis conjugation to a primary amino group thus an additional modification with an amino group is required. A C3, C6 or C12 amino group can be placed at the 5' or for the 3' end a C3 or C7 amino and for internal positions an amino modified base is used, e.g Amino dT C6.
Digoxigenin (as Digoxigenin-3-O-methylcarbonyl-epsilon-aminocaproic acid NHS ester) is a member of the steroid family found in Digitalis plants (1). It is a hapten, that is, a small molecule having high immunogenicity. Because antibodies raised against haptens have considerably higher affinities for them than other antibodies do for their targets makes haptens particularly desirable as affinity tags for oligonucleotides (2).
Digoxigenin ('Dig') is commonly used to label oligonucleotides probes for use in hybridization applications, for example, in situ hybridization (3), Northern and Southern blotting. After hybridization to their targets, these Dig-labeled probes are detected with anti-Dig antibodies that are labeled with dyes (for primary detection) or enzymes (for secondary detection using a fluorogenic, chemiluminogenic, or colorimetric substrate specific for the enzyme). To maximize signal, Gene Link recommends modifying the oligonucleotide probe with three or more Dig molecules, spaced about 10 bases apart. Note that since digoxigenin is in the form of an NHS ester, an active primary amino group (such as Amino Linker C6) must first be incorporated into the oligonucleotide, to allow for subsequent conjugation to the digoxigenin NHS ester.
References
1. Polya, G. Biochemical targets of plant bioactive compounds. New York: CRC Press, 2003. p 847.
2. Shreder, K. Synthetic Haptens as Probes of Antibody Response and Immunorecognition. Methods (Academic Press) (2000), 20: 372-379.
3. Hauptmann, G., Gerster, T.. Two-color whole-mount in situ hybridixation to vertebrate and Drosophila embryos. Trends Genet. (1994), 10: 266.
- Digoxigenin NHS
|
|
