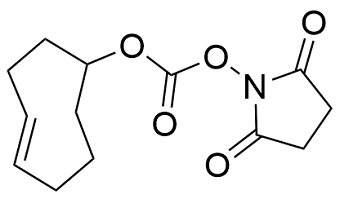
Modification : TCO NHS
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6756
Click Chemistry
[TCO-N]
Y
Y
Y
153.21
-
PS26-6756.pdf
-
-
-
| Catalog No | Scale | Price |
| 26-6756-05 | 50 nmol | $308.00 |
| 26-6756-02 | 200 nmol | $308.00 |
| 26-6756-01 | 1 umol | $394.00 |
| 26-6756-03 | 2 umol | $405.00 |
| 26-6756-06 | 5 umol | $1,773.00 |
| 26-6756-10 | 10 umol | $1,348.00 |
| 26-6756-15 | 15 umol | $1,685.00 |
This modification is a post synthesis conjugation to a primary amino group thus an additional modification with an amino group is required. A C3, C6 or C12 amino group can be placed at the 5' or for the 3' end a C3 or C7 amino and for internal positions an amino modified base is used, e.g Amino dT C6.
Click here for a complete list of Click Chemistry Oligo Modifications
TCO (trans-cyclooctene) NHS ester can be used to introduce an active azide group to an amino-modified oligonucleotide. Introduction can be done at either the 5'- or 3'-end, or internally. To do this, the oligo first must be synthesized with a primary amino functional group modification, e.g Amino C3, C6 or C12 for the 5' end or amino C3, C6 or C7 for the 3' end for the ends) or the amino C6 version of the base phosphoramidite (for internal labeling). The Azide C2 NHS ester is then manually attached to the oligo through the amino group in a separate reaction post-synthesis.
The presence of the azide allows the user to use "Click Chemistry" (a [3+2] cycloaddition reaction between alkynes and azides, using copper (I) iodide as a catalyst) to conjugate the azide-modified oligo to a terminal alkyne-modified oligo with extremely high regioselectivity and efficiency (1,2). Preparation of the alkyne-modified oligo can be achieved using the 5'-Hexynyl modifier (see its respective tech sheet for details). Click chemistry can be used to form short, cyclic oligos that can be used as research tools in various biophysical and biological studies (3). In particular, they have considerable potential for in vivo work, as cyclic oligos are known to be very stable in serum for up to several days.
 References
References
1. Huisgen, R.
Angew. Chem. Int. Ed. (1963),
2: 565-568.
2. Rostovtsev, V.V., Green, L.G., Fokin, V.V., Sharpless, K.B. A Stepwise Huisgen Cycloaddition Process: Copper(I)-Catalyzed Regioselective Ligation of Azides and Terminal Alkynes.
Angew. Chem. Int. Ed. (2002),
41: 2596-2599.
3. Kumar, R., El-Sagheer, A., Tumpane, J., Lincoln, P., Wilhelmsson, L.M., Brown, T. Template-Directed Oligonucleotide Strand Ligation, Covalent Intramolecular DNA Circularization and Catenation Using Click Chemistry.
J. Am. Chem. Soc. (2007),
129: 6859-6864.
- TCO NHS Oligo
