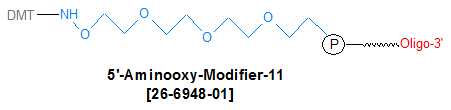
Modification : Aminooxy-Modifier-11
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6948
Conjugation Chemistry
[AmOx]
Y
N
N
271.21
-
PS26-6948.pdf
-
-
-
| Catalog No | Scale | Price |
| 26-6948-05 | 50 nmol | $343.00 |
| 26-6948-02 | 200 nmol | $343.00 |
| 26-6948-01 | 1 umol | $446.00 |
| 26-6948-03 | 2 umol | $670.00 |
| 26-6948-06 | 5 umol | $2,007.00 |
| 26-6948-10 | 10 umol | $3,572.00 |
| 26-6948-15 | 15 umol | $4,465.00 |
| Discounts are available for Aminooxy-Modifier-11! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
Oligos synthesized with Aminooxy Modifier 11 is supplied with 5'-DMT group to be removed at the time of reaction and facilitate reaction with aldehyde and ketones. See protocol below.
The 5'-AminoOxy-Modifier 11 contains the tetraethylene glycol linkage for improved solubility and minimizes the effects on hybridization of the oligo. Aminooxy modifiers can be used in hemoselective conjugation reactions with aldehydes and ketones to form stable oximes. The oxime formed from the reaction of these alkyloxyamines creates a stable covalent bond that is compatible with standard oligonucleotide deprotection conditions. Oxime covalent bonds are a more stable than the imine formed by the conjugation of amines with aldehydes, which require subsequent reduction to secondary amines.
Solution Phase Conjugation
Solution-phase with direct-conjugation to an aldehyde
or ketone provides the best results. Synthesize the oligo
DMT-ON and complete the deprotection. After drying, complete the conjugation in 80%
acetic acid to simultaneously remove the DMT group
and catalyze the oxime formation.
Solid Phase Conjugation
The aminooxy conjugation can also be performed on the
column after removal of the 5'-DMT group, provided the
label is stable to the subsequent deprotection conditions.
If the oligo will not be conjugated immediately, retain
the DMT protecting group and remove with 3% TCA in
DCM just prior to the conjugation.
Protocol: Conjugation reaction of 5' aminoxy modifier 11 oligo with aldehyde
Gene Link provides the oligo in a dried lyophilized form. The 5' aminoxy modifier 11 oligo is provided in a protected state with a DMT group at the 5' end. This is removed prior to the conjugation reaction with aldehyde by following the protocol below.
1. Suspend lyophilized oligo in water or in a concentration and volume as desired. An aliquot can be used for the conjugation reaction protocol.
2. Add 10 equivalents of aldehyde in a suitable solvent. Example. Total volume of 300 uL.
3. Add 100 uL of acetic acid. Vortex.
4. Incubate for 1 hour at room temperature.
5. Desalt and concentrate by ethanol precipitation protocol below or follow other purification protocol.
5. Add 50 uL of 3M Sodium Acetate and 1 mL of absolute ethanol.
6. Incubate at -20 C for 20 minutes.
7. Centrifuge at 10K rpm for 5 minutes.
8. Decant ethanol carefully so as not to dislodge the oligo pellet.
9. Dry pellet at room temperature.
- Aminooxy-Modifier-11
