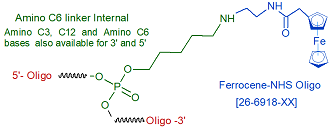
Modification : Ferrocene-NHS
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6918
Redox Electrochemical
[Fc-N]
Y
Y
Y
694.53
8.7
PS26-6906.pdf
-
-
-
| Catalog No | Scale | Price |
| 26-6918-05 | 50 nmol | $499.00 |
| 26-6918-02 | 200 nmol | $499.00 |
| 26-6918-01 | 1 umol | $588.00 |
| 26-6918-03 | 2 umol | $906.00 |
| 26-6918-06 | 5 umol | $1,812.00 |
| 26-6918-10 | 10 umol | $3,261.00 |
| 26-6918-15 | 15 umol | $4,565.00 |
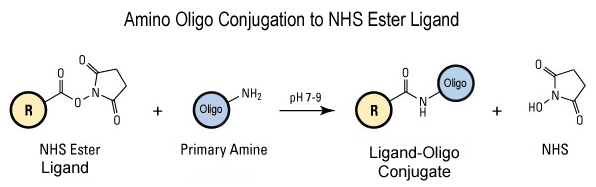
This modification is a post synthesis conjugation to a primary amino group thus an additional modification with an amino group is required. A C3, C6 or C12 amino group can be placed at the 5' or for the 3' end a C3 or C7 amino and for internal positions an amino modified base is used, e.g Amino dT C6.
Ferrocene oligonucleotides should be stored under Argon and aqueous solutions should be degassed immediately. A convenient way to degas is the use of vacuum desiccator. We suggest making multiple small aliquots for storage at -20C or -80C for long term storage.
Ferrocene is a redox-active ferrocene moiety. Ferrocene is a sandwich compound composed of two cyclopentadienyl rings bound on opposite sides of a central iron atom (1). When incorporated into an oligonucleotide, the presence of ferrocene enables its use as an electrochemical (EC) probe for nucleic acid analysis. Ferrocene-modified probes can be designed to bind to either single- or double-stranded targets, and the resulting double- or triple-stranded probe-target complex is typically detected by HPLC with a standard electrochemical detector, with reported sensitivity at the sub-femtomole level (2,3). Ferrocence-modified probes covalently attached to a gold electrode surface have also been used in EC-based SNP assay, one probe to detect wild-type, and the other the SNP (4). In an alternative format, a "sandwich SNP assay" has also been studied. Here, a capture oligo was covalently bound to a gold surface via several phosphorothiolate linkages to capture the desired target DNA and hold it close to the gold surface. The targeted region for the capture oligo contains the SNP. A second, ferrocene-modified detection probe, hybridizes to a different, highly conserved, part of the target oligo to serve as the detector. If the target has been captured, electron transfer occurs between the ferrocene of the detection probe and the gold surface, producing an electrochemical signal (5). Ferrocene-modified DNA aptamers, designed to bind to one specific biochemical target molecule (DNA, RNA, proteins, etc.) have also been used to make aptamer-based EC sensors (6). EC probes also have significant potential as a low cost alternative to fluorescent-based probes in DNA microarray systems designed for use in clinical or medical diagnosis (7,8).
-
References
1. Neto, F., Pelegrino, A., Caramori, A., Darin, V.A. Ferrocene: 50 Years of Transition Metal Organometallic Chemistry—From Organic and Inorganic to Supramolecular Chemistry. ChemInform (2004), 35: no. doi: 10.1002/chin.200443242.
2. Takenaka, S., Uto, Y., Kondo, H., Ihara, T., Takagi, M. Electrochemically active DNA probes: Detection of target DNA sequences at femtomole level by high-performance liquid chromatography with electrochemical detection. Anal. Biochem. (1994), 218: 436-443.
3. Ihara, T., Maruo, Y., Takenaka, S., Takagi, M. Ferrocene-oligonucleotide conjugates for electrochemical probing of DNA.Nucleic Acids Res. (1996), 24: 4273-4280.
4. Yu, C.J., Wan, Y., Yowanto, H., Li, J., Tao, C., James, M.D., Tan, C.L., Blackburn, G.F., Meade, T.J. Electronic Detection of Single-Base Mismatches in DNA with Ferrocene-Modified Probes. J. Am. Chem. Soc. (2001), 123: 11155-11161.
5. Nakayama, N., Ihara, T., Nakano, K., Maeda, M. DNA sensors using a ferrocene-oligonucleotide conjugate. Talanta (2002), 56: 857-866.
6. Radi, A-E., Sanchez, J.L.A., Baldrich, E., O’Sullivan, C.K. Reagentless, Reusable, Ultrasensitive Electrochemical Molecular Beacon Aptasensor. J. Am. Chem. Soc. (2006), 128: 117-124.
7. Liepold, P., Wieder, H., Hillebrandt, H., Friebel, A., Hartwich, G. DNA-arrays with electrical detection: A label-free low cost technology for routine use in life sciences and diagnostics.Bioelectrochem. (2005), 67: 143-150.
8. Liepold, P., Kratzmuller, T., Persike, N., Bandilla, M., Hinz, M., Wieder, H., Hillebrandt, H., Ferrer, E., Hartwich, G. Electrically detected displacement assay (EDDA): a practical approach to nucleic acid testing in clinical or medical diagnosis. Anal. Bioanal. Chem. (2008), 391: 1759-1772.
- Ferrocene-NHS
