In this section: Introduction | Quality Control | Purification | Modifications | Long Oligos | Price List
In this section: Introduction | Molecular Beacon FAQ's | Fluorescent Probes Price List | Other Fluorescent Molecular Probes
In this section: SPCT | DME SPCT Intro | Order DME TaqMan® Assays SPCT | SNP PCT Search | Gene Expression Assays | SPCT Design Center | GeneAssays
In this section: RNA Oligonucleotides | Quality Control | Purification | Modifications | RNAi Explorer™ Products and Prices | Custom RNAi | RNAi Design Guidelines | SmartBase™ siRNA Modifications | shRNA Explorer™
In this section: PCR Amplification & Analysis
In this section: Introduction | Genemer™ | GeneProber™ | Prober™ Gene Detection Kits | GScan™ Gene Detection Kits | Genemer™ Control DNA | Infectious Diseases
In this section: Gene Construction
In this section: Introduction | The Omni-Clean™ System | The Omni-Pure™ Plasmid Purification System | The Omni-Pure™ Genomic DNA Purification System | Viral DNA & RNA Purification | Microbial DNA Purification | Plant DNA Purification
In this section: Introduction | Quality Control | Purification | Modifications | Long Oligos | Price List
In this section: Introduction | Molecular Beacon FAQ's | Fluorescent Probes Price List | Other Fluorescent Molecular Probes
In this section: SPCT | DME SPCT Intro | Order DME TaqMan® Assays SPCT | SNP PCT Search | Gene Expression Assays | SPCT Design Center | GeneAssays
In this section: RNA Oligonucleotides | Quality Control | Purification | Modifications | RNAi Explorer™ Products and Prices | Custom RNAi | RNAi Design Guidelines | SmartBase™ siRNA Modifications | shRNA Explorer™
In this section: PCR Amplification & Analysis
In this section: Introduction | Genemer™ | GeneProber™ | Prober™ Gene Detection Kits | GScan™ Gene Detection Kits | Genemer™ Control DNA | Infectious Diseases
In this section: Gene Construction
In this section: Introduction | The Omni-Clean™ System | The Omni-Pure™ Plasmid Purification System | The Omni-Pure™ Genomic DNA Purification System | Viral DNA & RNA Purification | Microbial DNA Purification | Plant DNA Purification
2'-Amino-U
2'-Amino-U
Code : [2-AmU]
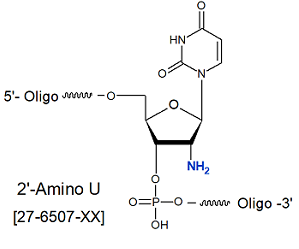
Modification : 2'-Amino-U
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
27-6507
Nuclease Resistance
[2-AmU]
Y
Y
Y
304.2
9.9
PS27-6507.pdf
260
-
-
| Catalog No | Scale | Price | | 27-6507-05 | 50 nmol | $270.00 | | 27-6507-02 | 200 nmol | $270.00 | | 27-6507-01 | 1 umol | $335.00 | | 27-6507-03 | 2 umol | $461.00 | | 27-6507-06 | 5 umol | $1,304.00 | | 27-6507-10 | 10 umol | $1,956.00 | | 27-6507-15 | 15 umol | $2,230.00 |
| Discounts are available for 2'-Amino-U! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
Oligonucleotide based diagnostics and therapy usually requires chemically modified bases for improved binding affinity/duplex stability, high selectivity and increased nuclease resistance.
The modification at 2' ribose position has shown to have stronger base-paring with target, which lead to increase duplex stability, enhanced specificity and nuclease resistance (1). These modifications includes 2'F, 2'O methyl, 2'O MOE, and 2' amino bases.
Libraries of RNA molecules containing 2'amino-(2′NH2)- or 2'fluoro-(2'F)-2'-deoxypyrimidines could yield ligands with similar nuclease resistance but not necessarily with similar affinities. This is because the intramolecular helices containing 2′NH2 have lower melting temperatures (Tm) compared with helices containing 2′F, giving them thermodynamically less stable structures and possibly weaker affinities. These were tested by isolating high-affinity ligands to human keratinocyte growth factor from libraries containing modified RNA molecules with either 2'-NH2 or 2'F pyrimidines. It was demonstrated that 2'F RNA ligands have affinities (Kd approximately 0.3-3 pM) and bioactivities (Ki approximately 34 pM) superior to 2'NH2 ligands (Kd approximately 400 pM and Ki approximately 10 nM). In addition, 2'F ligands have extreme thermostabilities (Tm approximately 78C in low salt (1).
Oligonucleotide modified with 2'-amino C and U bases has shown to increase half-life 10 times more than other modified base in serum stability. However binding capacity is not as strong as other modified base.
2'-Amino pyrimidine Applications
- Antisense Oligos
- Aptamers
- siRNA
ASO's and siRNA Modifications.
Click this link to view ASO's and siRNA Modifications.
ASO's and siRNA Delivery.
The development of effective delivery systems for antisense oligonucleotides is essential for their clinical therapeutic application. The most common delivery system involves a relatively hydrophobic molecule that can cross the lipid membrane. Cholesterol TEG, alpha-Tocopherol TEG ( a natural isomer of vitamin E), stearyl and GalNAc modifications have been shown to effective for delivery of ASO's and siRNA in addition to cell penetrating peptides.
Click this link to view these modifications.
REFERENCES:
1. Pagratis, N.C., et al., Potent 2'-amino- , and 2'-fluoro-2' deoxyribonucleotide RNA inhibitors of keratinocyte growth factor. (1997), 15: 68-73.
2. Lou, C. et al., Oligonucleotides Containing Aminated 2'-Amino-LNA Nucleotides: Synthesis and Strong Binding to Complementary DNA and RNA. Bioconjugate Chem. (2017). 28: 1214-1220 - 2'-Amino-U
|
|
