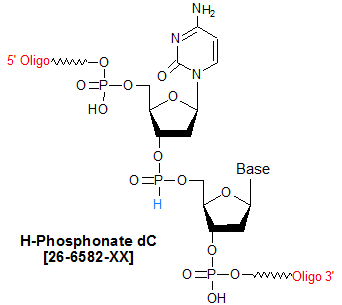
Modification : H-Phosphonate dC. dC(H-p)
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
| Catalog No | Scale | Price |
| 26-6582-05 | 50 nmol | $65.00 |
| 26-6582-02 | 200 nmol | $65.00 |
| 26-6582-01 | 1 umol | $84.00 |
| 26-6582-03 | 2 umol | $126.00 |
| 26-6582-06 | 5 umol | $378.00 |
| 26-6582-10 | 10 umol | $674.00 |
| 26-6582-15 | 15 umol | $842.00 |
H-Phosphonate modification has a setup charge of $250.00 per order for special synthesis reagents .
Special notes on H-phosphonate modification.
1. H-phosphonate chimeric oligos. H-phosphonate bases/oligos can be synthesized in a sequence with standard bases or modifications containing phosphodiester linkages the 3' end of the sequence. If a chimeric oligo is designed then the H-phosphonate should be on the 5' end with no other standard phosphodiester bases at 5' of the H-phosphonate linked bases. Chemical oligo synthesis starts from the 3' and the phosphodiester linkages are generated by oxidation. Any such oxidation cycle after coupling the H-phosphonate will oxidize the H-phosphonate linkage to phosphodiester linkage.
2. Special oxidation. H-phosphonate modified oligos can be supplied with oligo bound to CPG solid support for the end user to perform special oxidation at the H-phosphonate linkage. Example is for S-35 radio-labeled phosphorothioate linkage. Please specify when placing an order.
H-phosphonoamidites are deoxynucleoside amidites modified such that, when incorporated into an oligonucleotide, that base position will have an H-phosphonate backbone linkage instead of the standard phosphodiester linkage. In the H-phosphonate linkage, one of the oxygen bound to the phosphorus atom in a phosphodiester linkage has been replaced by hydrogen. H-phosphonate-modified oligos are typically synthesized when a researcher wants to prepare a S-35 radiolabeled phosphorothioate linkage (1). For the radiolabeled phosphorothioate, an H-phosphonate linkage is incorporated at the desired position using phosphoramidite chemistry, and then S-35 is used to replace the hydrogen atom via a sulfurization reaction (2). For the phosphoroamidate, N,N-dimethylaminoethylamine is reacted with the H-phosphonate to form the substituted linkage. Phosphoroamidates, being a cationic linkage, may provide nuclease resistance and improved cell permeability (3).
References
1. Maier, M.A., Guzaev, A.P., Manoharan, M. Synthesis of Chimeric Oligonucleotides Containing Phosphodiester, Phosphorothioate, and Phosphoramidate Linkages.
Org. Lett. (2000),
2: 1819-1822.
2. Wallin, R., Kalek, M., Bartoszewicz, A., Thelin, M., Stawinski, J. On the Sulfurization of H-Phosphonate Diesters and Phosphite Triesters Using Elemental Sulfur.
Phosphorus, Sulfur, and Silicon (2009),
184: 908-916.
3. Letsinger, R.L., Singman, C.N., Histand, G., Salunkhe, M. Cationic Oligonucleotides.
J. Am. Chem. Soc. (1988),
1110: 4470-4471.
- H-Phosphonate dC. dC(H-p)
