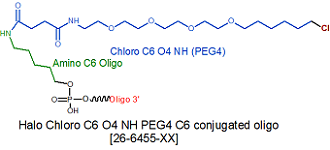
Modification : Halo Chloro Tag C6 PEG4 NHS
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6455
Others
[Halo-Cl-PEG4-N]
Y
Y
Y
394.91
-
PS26-6455.pdf
-
-
-
| Catalog No | Scale | Price |
| 26-6455-05 | 50 nmol | $810.00 |
| 26-6455-02 | 200 nmol | $810.00 |
| 26-6455-01 | 1 umol | $960.00 |
| 26-6455-03 | 2 umol | $1,188.00 |
| 26-6455-06 | 5 umol | $3,645.00 |
| 26-6455-10 | 10 umol | $4,752.00 |
| 26-6455-15 | 15 umol | $5,702.00 |
Halo Chloro Tag C6 PEG4 oligo conjugation is performed post synthesis of an oligo utilizing an amino group at the site for Halo ligand conjugation. A C3, C6 or C12 amino group can be placed at the 5' or for the 3' end a C3 or C7 amino and for internal positions an amino modified base is used, e.g Amino dT C6.
Halotag Protein Oligo Conjugation
The strategy of small-molecule fluorescent labeling of genetically encoded proteins has become a popular alternative to GFP labeling.
Among the most widely used approaches is the HaloTag method developed by Promega, which utilizes a bacterial haloalkane dehalogenase. The enzyme removes halides from aliphatic hydrocarbons by a nucleophilic displacement mechanism to form a covalent ester linkage between the haloalkane and Asp106 in the enzyme. In the wild type haloalkane dehalogenase, the ester is quickly hydrolyzed by histidine 272 in the catalytic active site. However, by mutating the histidine to phenylalanine, the HaloTag variant renders the covalent ester bond stable toward hydrolysis.
Further Reading and Protocols
HaloTag Ligand Building Blocks.
Halotag Protein Conjugation
3. 1. Los, G. V.; Encell, L. P.; McDougall, M. G.; Hartzell, D. D.; Karassina, N.; Zimprich, C.; Wood, M. G.; Learish, R.; Ohana, R. F.; Urh, M.; Simpson, D.; Mendez, J.; Zimmerman, K.; Otto, P.; Vidugiris, G.; Zhu, J.; Darzins, A.; Klaubert, D. H.; Bulleit, R. F.; Wood, K. V. HaloTag: a novel protein labeling technology for cell imaging and protein analysis. ACS Chem. Biol., 2008, 3, 373-382
4. Vijay Singh, Shenliang Wang, and Eric T. Kool, Genetically Encoded Multispectral Labeling of Proteins with Polyfluorophores on a DNA Backbone. J. Am. Chem. Soc., 2013, 16, 6184-6191.
- Halo Chloro Oligo Tag PEG4 NHS
