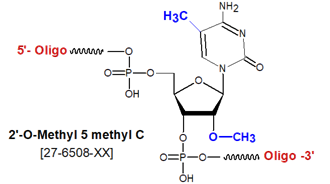
Modification : 2'-O Me-5-Me-C
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
27-6508
Structural Studies
[m5mC]
Y
Y
Y
333.24
7.4
PS27-6508.pdf
-
-
-
| Catalog No | Scale | Price |
| 27-6508-05 | 50 nmol | $124.00 |
| 27-6508-05 | 50 nmol | $142.00 |
| 27-6508-02 | 200 nmol | $142.00 |
| 27-6508-02 | 200 nmol | $124.00 |
| 27-6508-01 | 1 umol | $316.00 |
| 27-6508-01 | 1 umol | $168.00 |
| 27-6508-03 | 2 umol | $235.00 |
| 27-6508-03 | 2 umol | $241.80 |
| 27-6508-06 | 5 umol | $398.00 |
| 27-6508-10 | 10 umol | $815.00 |
| 27-6508-10 | 10 umol | $1,289.60 |
| 27-6508-15 | 15 umol | $1,612.00 |
| 27-6508-15 | 15 umol | $956.00 |
| Discounts are available for 2'-O Me-5-Me-C! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
Antisense Oligos (ODN) & siRNA Oligo Modifications
Click here for more information on antisense modifications, design & applications.
2'-OMethyl-5-methyl cytosine (2'-OMe-5-Me-C) is an RNA monomer that pairs with G, and when substituted for C in an oligonucleotide, both increases the stability of the resulting duplex relative to the comparable unmodified form, and confers nuclease resistance at that position(1). This "double-methylated"-modified cytosine thus is an excellent choice for incorporation into anti-sense oligos, where both properties are particularly desirable. Furthermore, because anti-sense oligonucleotides containing a CpG motif are known to induce pro-inflammatory responses after in vivo administration to animals, including human, via activation of Toll-like receptor 9 (TLR9), substitution of 2'-OMe-5-Me-dC for C in these motifs can prevent or sharply reduce these undesirable immune responses (2,3).
Modifications Increasing Duplex Stability and Nuclease Resistance
|
Modification
|
Duplex Stability [Tm Increase]
|
Nuclease Resistance
|
| Locked Analog Bases |
Increased [2- 4C per substitution] |
Increased |
| 2-Amino-dA |
Increased [3.0C per substitution] |
Similar to DNA |
| C-5 propynyl-C |
Increased [2.8C per substitution] |
Increased |
| C-5 propynyl-U |
Increased [1.7C per substitution] |
Increased |
| 2'-Fluoro |
Increased [1.8C per substitution] |
Increased |
| 5-Methyl-dC |
Increased [1.3C per substitution] |
Similar to DNA |
| 2'-O Methyl |
Increased |
Increased |
| Phosphorothioate |
Slightly decreased |
Increased |
|
Click here for complete list of duplex stability modifications
|
References
1. Bundock, P.; de Both, M.T.J.; Hogers, R.C.J. 2006. Alternative nucleotides for improved targeted nucleotide exchange. Patent No. 2007073149, filed Dec 22. 2005, issued June 28, 2007.
2. Henry, S.P.; Stecker, K.; Brooks, D.; Monteith, D.; Conklin, B.; Bennett, C.F. Chemically modified oligonucleotides exhibit decreased immune stimulation in mice.
J. Pharmacol. Exp. Ther. (2000),
292: 468-479.
3. Yu, D.; Wang, D.; Zhu, F.-G.; Bhagat, L.; Dai, M.; Kandimalia, E.R.; Agrawal, S. Modifications Incorporated in CpG Motifs of Oligodeoxynucleotides Lead to Antagonist Activity of Toll-like Receptors 7 and 9.
J. Med. Chem. (2009),
52: 5108-5114.
- 2'-O Me-5-Me-C
