In this section: Introduction | Quality Control | Purification | Modifications | Long Oligos | Price List
In this section: Introduction | Molecular Beacon FAQ's | Fluorescent Probes Price List | Other Fluorescent Molecular Probes
In this section: SPCT | DME SPCT Intro | Order DME TaqMan® Assays SPCT | SNP PCT Search | Gene Expression Assays | SPCT Design Center | GeneAssays
In this section: RNA Oligonucleotides | Quality Control | Purification | Modifications | RNAi Explorer™ Products and Prices | Custom RNAi | RNAi Design Guidelines | SmartBase™ siRNA Modifications | shRNA Explorer™
In this section: PCR Amplification & Analysis
In this section: Introduction | Genemer™ | GeneProber™ | Prober™ Gene Detection Kits | GScan™ Gene Detection Kits | Genemer™ Control DNA | Infectious Diseases
In this section: Gene Construction
In this section: Introduction | The Omni-Clean™ System | The Omni-Pure™ Plasmid Purification System | The Omni-Pure™ Genomic DNA Purification System | Viral DNA & RNA Purification | Microbial DNA Purification | Plant DNA Purification
In this section: Introduction | Quality Control | Purification | Modifications | Long Oligos | Price List
In this section: Introduction | Molecular Beacon FAQ's | Fluorescent Probes Price List | Other Fluorescent Molecular Probes
In this section: SPCT | DME SPCT Intro | Order DME TaqMan® Assays SPCT | SNP PCT Search | Gene Expression Assays | SPCT Design Center | GeneAssays
In this section: RNA Oligonucleotides | Quality Control | Purification | Modifications | RNAi Explorer™ Products and Prices | Custom RNAi | RNAi Design Guidelines | SmartBase™ siRNA Modifications | shRNA Explorer™
In this section: PCR Amplification & Analysis
In this section: Introduction | Genemer™ | GeneProber™ | Prober™ Gene Detection Kits | GScan™ Gene Detection Kits | Genemer™ Control DNA | Infectious Diseases
In this section: Gene Construction
In this section: Introduction | The Omni-Clean™ System | The Omni-Pure™ Plasmid Purification System | The Omni-Pure™ Genomic DNA Purification System | Viral DNA & RNA Purification | Microbial DNA Purification | Plant DNA Purification
Atto 465 NHS
Atto 465-N
Code : [Atto465-N]
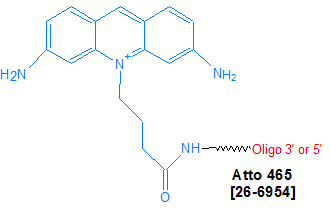
Modification : Atto 465-N
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6954
Fluorescent Dyes
[Atto465-N]
Y
Y
Y
493
-
PS26-6954.pdf
453
508
-
| Catalog No | Scale | Price | | 26-6954-05 | 50 nmol | $702.00 | | 26-6954-02 | 200 nmol | $702.00 | | 26-6954-01 | 1 umol | $913.00 | | 26-6954-03 | 2 umol | $2,754.00 | | 26-6954-06 | 5 umol | $4,108.50 | | 26-6954-10 | 10 umol | $7,301.00 | | 26-6954-15 | 15 umol | $9,126.00 |
Click here for a list of fluorophores.
This modification is a post synthesis conjugation to a primary amino group thus an additional modification with an amino group is required. A C3, C6 or C12 amino group can be placed at the 5' or for the 3' end a C3 or C7 amino and for internal positions an amino modified base is used, e.g Amino dT C6.
Conventional and popular dyes that are derivatives of fluoroscein (FAM, HEX and TET) and Cyanine dye derivatives (Cy3, Cy5, Cy5.5, Cy7 etc) are commonly used for fluorescently labeling oligos for use as molecular probes for real time PCR, FISH analysis and fragment analysis. For most purposes these provide a good range in wavelength and other optical properties and are available as amidites for direct coupling to oligos using automated chemistry. Other fluorescent dyes are available as N-hydroxysuccinimide (NHS) for conjugation using a primary amine group linked to the oligos. A new series of Atto dyes are now available that are are designed for high sensitivity applications, including single-molecule detection.
ATTO Dyes are a series of fluorescent labels and dyes manufactured by ATTO-TEC GmbH in Siegen, Germany. The ATTO Dye series covers a spectral range from 390 nm in the UV to 740 nm in the near infrared allowing excitation with most commonly used light sources. The dyes typically are derivatives of coumarins, rhodamines, carbopyronins and oxazines.
Compared with other labels especially for the red region of the spectrum, ATTO-labels show excellent photostability and brightness. Atto labels have rigid structures that do not show any cis-trans isomerization. Thus these labels display exceptional intensity with minimal spectral shift on conjugation. The molecules of most common dyes, e.g. cyanines, have a more or less flexible structure. Hence their solutions contain a mixture of several isomers with varying properties. Since the equilibrium between the isomers depends on temperature and other environmental factors, absorption and fluorescence of such dyes are ill-defined. ATTO-dyes have a molecular structure that ensures high rigidity of the chromophore. They do not form equilibria with various isomers, their optical properties are nearly independent of solvent and temperature. ATTO 647N fluoresces twice as strong as Cy5 in aqueous solution. In addition many common fluorescent labels especially cyanine dyes like Cy5 deteriorate even without any irradiation (in the dark), in particular when exposed to small concentrations of ozone present in the laboratory atmosphere. Under identical conditions of ozone exposure the new dyes ATTO 633, ATTO 647N and ATTO 655 last up to 100 times longer than cyanines like Cy5 and Alexa Fluor 647. This is very important in microarray applications, where the dye molecules are located at the surface and thus are in direct contact with the atmosphere.
- Atto 465 NHS
|
|
