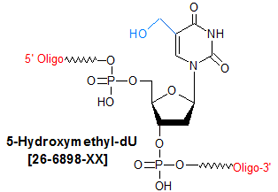
Modification : 5-hm dU
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
| Catalog No | Scale | Price |
| 26-6898-05 | 50 nmol | $337.00 |
| 26-6898-02 | 200 nmol | $337.00 |
| 26-6898-01 | 1 umol | $447.00 |
| 26-6898-03 | 2 umol | $521.00 |
| 26-6898-06 | 5 umol | $861.50 |
| 26-6898-10 | 10 umol | $1,180.00 |
| 26-6898-15 | 15 umol | $1,775.00 |
| Discounts are available for 5-hm dU! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
5-Hydroxymethyl deoxyuridine (5hmdU; 5-hm-dU) is a minor DNA base; its presence in DNA strands occurs by either oxidative attack via peroxide radicals, or ionizing radiation, on the 5-methyl group of thymine (1,2). Available evidence does not support 5-hm-dU being mutagenic; however, base excision repair enzymes specific to it (i.e., hydroxymethyluracil-DNA glycosylases) are known to exist in protists and animals (3), suggesting that this lesion nevertheless may have mutagenic potential. Incorporation of 5-hm-dU into synthetic oligos for use in studie into the molecular genetics and enzymology of DNA base excision repair pathways.
However, because 5-hm-dU also appears as a deamination intermediate during the oxidative de-methylation of 5-methyl-dC to dC, 5-hm-dU can be used in studies into the role of 5-methyl-dC de-methylation in epigenetic regulation.
References
1. Cadet. J., Berger, M., Douki, T., Ravanat, J-L. Oxidative damage to DNA : formation, measurement, and biological significance.
Rev. Physiol. Biochem. Pharmacol. (1997),
131: 1-87.
2. Teebor, G.W., Frenkel, K., Goldstein, M.S. Ionizing radiation and tritium transmutation both cause formation of 5-hydroxymethyl-2’-deoxyuridine in cellular DNA.
Proc. Natl. Acad. Sci. USA. (1984),
81: 318-321.
3. Levy, D.D., Teebor, G.W. Site directed substitution of 5-hydroxymethyluracil for thymine in replicating phiX-174am3 DNA via synthesis of 5-hydroxymethyl-2’-deoxyuridine-5’-triphosphate.
Nucleic Acids Res. (1991),
19: 3337-3343.
- 5-Hydroxymethyl-dU
