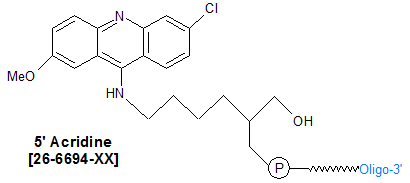
Modification : Acridine
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6694
Intercalators
[Acrd]
Y
Y
Y
450.86
39.5
PS26-6694.pdf
-
-
-
| Catalog No | Scale | Price |
| 26-6694-05 | 50 nmol | $350.00 |
| 26-6694-02 | 200 nmol | $350.00 |
| 26-6694-01 | 1 umol | $456.00 |
| 26-6694-03 | 2 umol | $683.00 |
| 26-6694-06 | 5 umol | $2,052.00 |
| 26-6694-10 | 10 umol | $3,645.00 |
| 26-6694-15 | 15 umol | $4,556.00 |
| Discounts are available for Acridine ! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
Acridine is classified as a DNA intercalating agent. Labeling of DNA oligos with acridine allows them to rapidly and stably intercalate into a target dsDNA molecule, adding increased stability to the double helix (1). In addition, incorporation of acridine to the 3'-end of an oligo confers a high level of exonuclease resistance to that end (2). Thus, oligos labeled with acridine may be useful in applications involving oligo hybrids requiring higher levels of stability.
Acridine-labeled oligos containing a polypyrimidine sequence possess the ability to form triplex helices that are highly stable, and, due to their increased hydrophobicity, can pass through membranes more easily than normal oligos. Such oligos were used as anti-sense reagents to suppress c-myc protooncogene expression and control tumor growth in mice (3), and as a transcriptional repressor to the IL-2 Receptor (4).
Since acridine is a fluorescent dye, it can also be use to make dye-labeled oligos. An interesting such application is the use of acridine-labeled primers to study non-enzymatic-template-directed RNA synthesis to provide experimental support for theories concerning possible replication of genetic information by early life forms on Earth (5).
References
1. Fukui, K., Tanaka, K. The Acridine Ring Selectively Intercalated into a DNA Helix at Various Types of Abasic Sites: Double Strand Formation and Photophysical Properties.
Nucleic Acids Res. (1996),
24: 3962-3967.
2. Gamper, H.B., Reed, M.W., Cox, T., Virosco, J.S., Adams, A.D., Gall, A.A., Scholler, J.K., Meyer, R.B. Facile preparation of nuclease resistant 3� modified oligodeoxynucleotides.
Nucleic Acids Res. (1993),
21: 145-150.
3. Stewart, D.A., Xu, Xiaohou, Thomas, S.D., Miller, D.M. Acridine-modified, clamp-forming antisense oligonucleotides synergize with cisplatin to inhibit c-Myc expression and B16-F0 tumor progression.
Nucleic Acids Res. (2002),
30: 2565-2574.
4. Grigoriev, M., Praseuth, D., Robin, P., Hemar, A., Saison-Behmoaras, T., Dautry-Varsat, A., Thuong, N.T., Helene, C., Harel-Bellan, A. A Triple-Helix-forming Oligonucleotide-Intercalator Conjugate Acts as Transcriptional Repressor via Inhibition of NFkB Binding to Interleukin-2 Receptor alpha-Regulatory Sequence.
J. Biol. Chem. (1992),
267: 3389-3395.
5. Kurz, M., Gobel, K., Hartel, C., Gobel, M.W. Acridine-labeled primers as tools for the study of nonenzymatic RNA oligomerization.
Helv. Chim. Acta (1998), 81: 1156-1180.
- Acridine
