In this section: Introduction | Quality Control | Purification | Modifications | Long Oligos | Price List
In this section: Introduction | Molecular Beacon FAQ's | Fluorescent Probes Price List | Other Fluorescent Molecular Probes
In this section: SPCT | DME SPCT Intro | Order DME TaqMan® Assays SPCT | SNP PCT Search | Gene Expression Assays | SPCT Design Center | GeneAssays
In this section: RNA Oligonucleotides | Quality Control | Purification | Modifications | RNAi Explorer™ Products and Prices | Custom RNAi | RNAi Design Guidelines | SmartBase™ siRNA Modifications | shRNA Explorer™
In this section: PCR Amplification & Analysis
In this section: Introduction | Genemer™ | GeneProber™ | Prober™ Gene Detection Kits | GScan™ Gene Detection Kits | Genemer™ Control DNA | Infectious Diseases
In this section: Gene Construction
In this section: Introduction | The Omni-Clean™ System | The Omni-Pure™ Plasmid Purification System | The Omni-Pure™ Genomic DNA Purification System | Viral DNA & RNA Purification | Microbial DNA Purification | Plant DNA Purification
In this section: Introduction | Quality Control | Purification | Modifications | Long Oligos | Price List
In this section: Introduction | Molecular Beacon FAQ's | Fluorescent Probes Price List | Other Fluorescent Molecular Probes
In this section: SPCT | DME SPCT Intro | Order DME TaqMan® Assays SPCT | SNP PCT Search | Gene Expression Assays | SPCT Design Center | GeneAssays
In this section: RNA Oligonucleotides | Quality Control | Purification | Modifications | RNAi Explorer™ Products and Prices | Custom RNAi | RNAi Design Guidelines | SmartBase™ siRNA Modifications | shRNA Explorer™
In this section: PCR Amplification & Analysis
In this section: Introduction | Genemer™ | GeneProber™ | Prober™ Gene Detection Kits | GScan™ Gene Detection Kits | Genemer™ Control DNA | Infectious Diseases
In this section: Gene Construction
In this section: Introduction | The Omni-Clean™ System | The Omni-Pure™ Plasmid Purification System | The Omni-Pure™ Genomic DNA Purification System | Viral DNA & RNA Purification | Microbial DNA Purification | Plant DNA Purification
NHS-Carboxy C10 5'
NHS-Carboxy C10 5'
Code : [NHS-CO-C10]
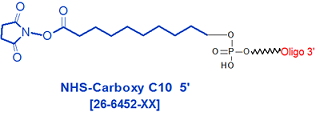
Modification : NHS-Carboxy C10 5'
Catalog Reference Number
Category
Modification Code
5 Prime
3 Prime
Internal
Molecular Weight (mw)
Extinction Coeficient (ec)
Technical Info (pdf)
Absorbance MAX
Emission MAX
Absorbance EC
26-6452
Conjugation Chemistry
[NHS-CO-C10]
Y
N
N
364.3
-
PS26-6452.pdf
-
-
-
| Catalog No | Scale | Price | | 26-6452-05 | 50 nmol | $212.00 | | 26-6452-02 | 200 nmol | $212.00 | | 26-6452-01 | 1 umol | $256.00 | | 26-6452-03 | 2 umol | $295.00 | | 26-6452-06 | 5 umol | $607.50 | | 26-6452-10 | 10 umol | $1,078.00 | | 26-6452-15 | 15 umol | $1,348.00 |
| Discounts are available for NHS-Carboxy C10 5'! |
| Modification* Discount Price Structure |
|
1 site/order
|
List price
|
|
2 sites/order
|
10% discount
|
|
3 sites/order
|
20% discount
|
|
4 sites/order
|
30% discount
|
|
5-9 sites/order
|
50% discount
|
|
10+ sites/order
|
60% discount
|
|
*Exceptions apply
|
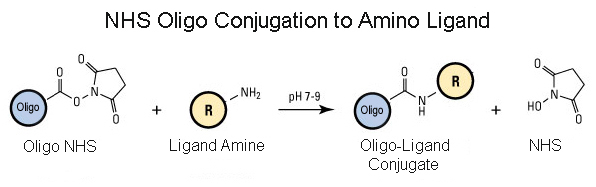
NHS-Carboxy-C10-modified oligos are supplied with oligo bound to CPG solid support for the end user to perform the amino ligand conjugation or Gene Link can conjugate the NHS-Oligo to user supplied or specified amino-ligand. See detailed protocol below.
Glen Report: NHS carboxy C10 & NHS Carboxy dT conjugation to amine ligands. NHS Carboxy Glen Report.
5'-Carboxy-Modifier C10 succinimidyl ester (NHS) can be used to incorporate an active carboxyl group onto the 5'-end of an oligonucleotide or an NHS group. The carboxyl group is separated from the 5'-end nucleotide base by a 10-carbon spacer arm to minimize steric interaction between the reactive group and the oligo. The presence of the carboxyl group allows the user to 5'-end label the oligo with a variety of different affinity, reporter or protein moieties (as primary amines/thiols or maleimides), depending on the application. Examples include biotin, digoxigenin, fluorescent dyes or quenchers, and enzymes (for example, alkaline phosphatase).
Carboxy C10 NHS protected carboxyl is supplied as oligo bound to CPG solid support and the 5' has an active NHS group. The important advantage of this NHS modification is that conjugation to the desired ligand is accomplished while the oligonucleotide is still attached to its solid support. This property enables the conjugation reaction to be carried out in an anhydrous organic solvent system, facilitating the linking of even very lipophilic moieties to oligos. In many cases, the presence of the lipophile also allows the lipophile-oligo conjugate to be efficiently purified by reverse-phase HPLC or reverse-phase cartridge.
Standard Conjugation Protocol Using Solid Phase NHS-Oligo-CPG
Ensure that the amino-ligand to be conjugated to the NHS-Oligo bound to CPG is stable to exposure to ammonium hydroxide solution (30%) for ~24 hrs. The ammonium hydroxide treatment is required to cleave the oligo from CPG bound oligo after the NHS conjugation is performed.
Gene Link provides custom oligo modified with NHS-carboxy C10 and NHS-Carboxy dT bound to CPG (controlled pore glass). The oligo is in a protected form and has to be deprotected and cleaved from the CPG after completion of the amino ligand conjugation to NHS.
The protocol given below has been tested with amino labeled dyes and other amino ligands soluble in DMSO and DMF.
1. NHS-oligo labels almost quantitatively and rapidly at room temperature. Keep reaction for 15 minutes or up to 3 hrs.
2. Split NHS-Oligo-CPG into two 2mL Screw Cap tubes. It is prudent to test one and then the second tube.
3. Should have at least 4 mg of amino ligand for 200 nmol scale synthesis oligo on CPG.
4. Prepare 200 uL of DMSO or DMF containing 1% diisopropylethylamine [CAS: 7087-68-5] or 10% Triethylamine [CAS: 121-44-8].
5. Dissolve ~4 mg amino-Ligand in 200 uL DMSO or DMF, containing 1% diisopropylethylamine [CAS: 7087-68-5] or 10% Triethylamine [CAS: 121-44-8].
6. Allow to react at room temperature for 15 to 30 minutes or longer. Vortexing intermittently.
7. Centrifuge at 3K rpm for 30 seconds.
8. Aspirate with a pipet unreacted ligand and save for future use.
9. Wash Oligo-CPG with 0.5 mL acetonitrile or DMF.
10. Repeat step 9 three times.
11. Dry CPG at room temperature. This is now labelled with the amino-ligand to the NHS group on the oligo bound to CPG.
12. Deprotect oligo and cleave from CPG by adding 1 mL cold 30% ammonium hydroxide (stored in a freezer) and keep 18- 24 hrs. at room temperature. It is essential to incubate in screw cap tubes with cap closed securely to prevent ammonia leakage. Vortex occasionally.
13. Open caps of the tube inside a hood and leave it for nearly 30 minutes for ammonia to evaporate.
14. Gently aspirate the ammonia solution using a pipet. Transfer to a fresh 2 mL screw cap tube.
15. Add 0.5 mL sterile water to the CPG, vortex gently and aspirate the solution using a pipet. Pool with the previous aspirated solution.
16. Dry the solution using a speedvac.
DNA Oligo Processing
Protocol steps 17 to 21 are ONLY for DNA oligos. See below for RNA Oligos processing. RNA oligos require special deprotection with TFAB.
17. Dissolve pellet in 0.4mL sterile water. Add 50 uL of 3 M Sodium Acetate pH 5.2 and 1 mL ethanol. Incubate at -20 C for 20 minutes.
18. Centrifuge at 12K rpm for 5 minute. Decant ethanol carefully so as not to disturb the pellet.
19. Add 0.5 mL 70% ethanol, vortex and centrifuge at 12K rpm for 5 minute.
20. Dry pellet at room temperature.
21. The pellet contains the oligo conjugated to your amino ligand. Process for purification if desired by HPLC or 7M urea-polyacrylamide gel.
RNA Oligo Processing.
Protocol after step 16 above. RNA oligos require special deprotection with TFAB.
RNA Oligos ONLY Processing. Follow strict RNase free protocol.
17. Add 100 uL DEPC water to the dried pellet from step 16. Vortex to partially dissolve.
18. Add 1 mL of 1M TBAF (tetrabutylammoniumfluoride) in THF [CAS Number: 429-41-4]. Vortex vigorously and ensure there are no visible pellets. Add more TBAF if required. Incubate overnight 12-16 hours at RT
19. Aliquot 200 uL TBAF RNA solution to each 1.5 mL tube.
20. To each tube add 1 mL 2% Lithium perchlorate [CAS Number: 7791-03-9] in acetone stored at -20oC. Vortex. Keep at -20oC for 30 minutes.
21. Spin at 10K rpm for 2 minute to collect the RNA pellet.
22. Wash pellet with 500 uL 70% Ethanol. Spin at 10K rpm for 2 minute to collect the RNA pellet.
23. Dissolve RNA in 400 uL DEPC water and perform ethanol precipitation.
24. Dissolve RNA oligo in 400 uL DEPC water and record A260 units to determine concentration.
25. Run gel or other method of quality control.
- NHS-Carboxy C10 5'
|
|
